How can we help you?
These are key areas that we can meet your expectation for our professional support.
- Support for Ingredients Check
- Support for Import Clearance Procedures
- Support for Finding Primary Distributor and Cooperator
TIPS
We are happy to share our POVs on cosmetic regulations in Japanese market with you to understand the essentials of its business environment for your preliminary study, if you are willing to bring your cosmetic or medicated cosmetic products into the market.
Just click the pdf icons below and read through the articles which have been re-edited from Mike Hayashida’s webinar and publication in the past for your further understanding.
TIPS-One

TIPS-Two
The Second Study

Q&A for TIPS
The following series of Q&As is based upon actual inquiries we have got from audiences of the Mike Hayashida’s webinar. If you have something else to come up with, please do not hesitate to add yours at the bottom of it!
- Q: How long does a registration of a cosmetic product take?
- A: The procedure of cosmetic products is not to obtain an approval of the government but to incur ‘submission’ to the government instead.
The government (MHLW) checks the formality of the submitted documents, and it usually takes approx. one week.
- Q: How long does a registration of a quasi-drug product take?
- A: Unlike cosmetic products I mentioned above, the procedure of quasi-drug products is to obtain an approval of the government, therefore, it at least takes half a year up to 5 years. If you challenge to register a new active ingredients for the first time in the market, it might actually take more like 10 or 15 years.
- Q: Can a foreign cosmetic manufacturer itself notify the products in Japan? Or does it need a responsible agent in Japan, who is handling all regulatory work on behalf of the foreign manufacturer?
- A: The most essential entity to sell foreign cosmetic products in Japan is a ‘primary distributor’ as your local counterparty in Japan. And the primary distributor is responsible for all the import operations including notification or registration to the Japanese government instead of the foreign manufacturer.
- Q: We assume that a foreign cosmetic manufacturer can only sell its products by a Japanese sales company. What are the responsibilities of this sales company? Does the sales company has to notify the products? Will the sales company be registration holder?
- A: Your assumption is almost correct, but the liable entity is a primary distributor not a seller. The primary distributor will notify to the government on behalf of all the stakeholders, as I mentioned above.
- Q: Can the registration licence be transferred to another company, once the registration has been successfully finished?
- A: The sales company does not have any license of selling, so that you cannot transfer it to another sales company.
Just let me re-emphasize that the key entity of our import and sales system is a primary distributor, not anyone else.
- Q.What is the limit of perfuming ingredient to be included in the quasi drug product?
- A: There’s no limit of perfuming ingredient in Q-D products.
- Q.What is the definition of Carry-over in Japan? Does it include the preservatives intentionally added, the antioxidant but also the impurities contained in the raw material (residual solvents, etc.)?
- A: In Japan, carry-over is defined as “the substances (including impurities) pertaining to the main ingredients and those not sufficient enough to perform the expected functions”. Therefore it includes everything such as preservatives、antioxidant、residual solvents, etc.
- Q.For Annex I (=list of forbidden substances), how to interpret this ban? Does it mean the substances have to be not detected in the finished product (so the limit acceptable will depend on the method used)? Is it up to the companies to establish their own acceptable limits for each substance? How the authorities interpret it?
- A: Yes, it mean the substances have to be not detected in the finished product. There’s no acceptable limits, but ‘zero’ is minimum requirement.
- Q.In Japan, the notion of traces technically unavoidable does not exist?
- A: In terms of the traces of ingredients, in Japan we have the concept called “carry-over”, which is defined as “the substances (including impurities) pertaining to the main ingredients and those not sufficient enough to perform the expected functions”, and it includes residual solvent. In Japan you do not have to show the carry-over ingredients on the label.
- Q.What other texts should be considered for cosmetic ingredients (except the Standards for cosmetic and texts listed in this standard)? Example is poisonous standard applicable for cosmetics/Quasi drugs?
- A: Aren’t Fig.12 or 13 sufficient for you?
- Q.Is it relevant to refer to the CLS nowadays as this text is not in force anymore?
- A: The CLS is no longer effective. However, the cosmetic industry tends to regard, on a tacit understanding, that it is better to manufacture the product based upon this standard because this standard can be a guideline for the product safety.
On the other hand, however, the competent authorities do not consider the CLS since it is no longer effective.
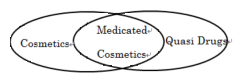
- Q.How to define a quasi-drugs, medicated cosmetic drugs, cosmetic? And how to differentiate them? For above 3 kind of products, what kind of documents does the government require before manufacture or sell the products?
- A: Aren’t Fig.2 and 3 sufficient to understand the difference?
- Q.Is there any limit for the new raw material? How to define a new raw material?
- A: No limit of raw material, as long as it’s not listed on a negative list.
- Q.Is there any website we can search to check the Japanese regulation of raw material? Like Cosing.
- A: https://www.cosmetic-info.jp/jcln/search.php
Although it’s written in Japanese, the above website might be your help
- Q.For QD license for manufacturing, 2 persons are required. Is it necessary to have at least one pharmacist or is it possible to have two people with any of the background explained during the webinar?
- A: No need to have a pharmacist, but It’s OK to have two people with any of the background introduced in the webinar.
- For the claims for the QD:
-
- Q.Claims will depend on the active(s) used or the category of QD? Is there an existing list of positive claims possible depending on the actives used in the formula or depending on the category of QD?
- A: Probably, no such a list available.
- Q.For claims associated with the formula what can we claim (56 cosmetic claims without any condition/other claims possible? If yes, what are the conditions to claim something out of the scope of the 56 cosmetic claims)?
- A: There should be no ‘out of the scope’ other than the 56 cosmetic claims since it’s a positive list.
- Q.Is the CLS (Comprehensive Licensing Standard of Cosmetic) could be used for a safety approach even if this text is no longer in force since the deregulation of cosmetic products? Is it considered by the competent authorities?
- A: The CLS is no longer effective. However, the cosmetic industry tends to regard, on a tacit understanding, that it is better to manufacture the product based upon this standard because this standard can be a guideline for the product safety.
On the other hand, however, the competent authorities do not consider the CLS since it is no longer effective.
- Q.What is the status of a personalized cosmetic product in Japan? Does the Japanese regulation authorize this kind of practices?
- A: As long as the product consists of four fundamental elements a) water, b) oil, c) base material and d) fragrance to be completed to perform as a cosmetic product, it’ll be OK’d.
- Q.How are toothpastes regulated in Japan? Are they considered cosmetics or medicated cosmetics? For example, in the US, if it contains fluoride for anti-cavity claims, they are considered to be drugs.
- A:It depends on what to claim. If it’s to ‘prevent tooth decay by whitening with brushing’, it’ll be categorized as ‘Cosmetics’. If it’s to ‘prevent pyorrhea by whitening with brushing ’, it’ll be categorized as ‘Q-Ds’. If it’s to ‘prevent periodontal diseases’, it’ll be categorized as ‘Drugs’.
- Q.If a cosmetic ingredient is NOT listed in the ENCS or ISHL inventories, can they be used in Japan? (As long as they are also not on the negative list?)
- A: Yes, they can.
- Q.We saw that ‘soap by sterilization’ was an example of a medicated cosmetic. Can you provide an example of such a product or the claim it would make? I thought soaps were considered cosmetic since they are used for cleansing the body.
- A: Let me show you one example of medicated soap categorized as Q-D.
http://www.musejapan.jp/
- Q.What does the application entail to gain approval to sell a cosmetic in Japan? Does it have to be submitted in Japanese?
- A: See fig.8. Yes, those application must be submitted in Japanese.
- Q.What safety testing and microbiological testing are required to launch a product in Japan?
- A: Japan Cosmetic Industry Association introduced the voluntary standards which conform to ISO 17516 (Microbiological Limits).
- Q.What are Biological Materials?
- A: MHLW Notification No. 210. Japanese Standards for Biological Ingredients regulates the biological materials, but English translation is not available. The following PDF may be helpful.
https://www.pmda.go.jp/files/000204341.pdf
- Q.What elements are mandatory on the product label?
- A: Please refer to JETRO document below, for description of Fair Competition Codes based upon Act against Unjustifiable Premiums and Misleading Representations regarding product labeling.
https://www.jetro.go.jp/ext_images/mexico/mercadeo/1Ecosme.pdf
- Q.How would you classify a body lotion, hand lotion or foot lotion? Would they all be cosmetics?
- A: Yes, they are all categorized as cosmetics.
- Q.Does an ingredient drive a medicated classification for a product? Or is it a combination of ingredient + claims?
- A: Ingredient alone drives the medicated classification.
- Q.Could you have a product without an active ingredient but strong claims which classifies the product as a drug?
- A: No, it must have an active ingredient.
- Q.I thought the PAL is already amended to PMD-Act in 2014 to safety measures on drugs and medical devices, and to revise better regulatory framework for medical devices. However, in the presentation it still used PAL. Which is the correct information?
- A: You are right, but nothing has amended what we presented.
- Q.In Fig.3, cosmetic has two categories:” cosmetic (general)” and “medicated cosmetic”, and “medicated cosmetic” is a part of quasi-drug. However I’ve learned that there are 3 three categories: drugs, quasi-drugs and cosmetics that related to beauty products. (see the link: http://www.mhlw.go.jp/english/wp/wp-hw2/part2/p3_0034.pdf ) That classification confuses me.
- A: Medicated cosmetics is a category in-between Cosmetics and Q-Ds. It’s just a matter of how I plotted the figure.
- Q.In Fig.22+23, it’s Drug>Medicated cosmetics>cosmetics, there is no mention of quasi-drug. How should I claim the product if it is a quasi-drug but not a medicated cosmetics?
- A: The outline of the regulation is the same, however, a category depends upon the product and its function that are quite diversified.
- Q.In Fig.16, There are 56 categories which are allowed as effectiveness in the case of cosmetic. No. 18 said “Prevent blemishes and rashes”. However, In Fig.23 said “prevent acne by facial wash (soap)”, which is more detailed than that in Fig18. This detailed effectiveness is also acceptable in Japan? Or this should be always the same as the word mentioned in平成12年12月28日付け厚生省医薬安全局長通知?
- A: A claim always have to be the same as it’s described in厚生省医薬安全局長通知
- Q.According to fig 4, the slide said the approval system is abolished in 2001, however, fig. 15 says “Time required for APPROVAL” is within 1 week. I would like to know in which point the approval system has been abolished and in which point the approval system has not been abolished. In Fig 6 it says Governmental approval is required for primary distributor and manufacturer, but not for seller. This is confusing me and it would be great if you can explain more in this point. I thought seller would need license.
- A: Sorry for my wrong use of the term ‘Approval’, but let me correct it to ‘ACCEPTANCE’ of submitted forms and documents. And a seller do not need license.
- Q.In slide 13, where is the list sourced from? And could you let me know the Japanese and English name of that law?
- A: If I was correct to get to your point, the negative list as I introduced in‘Fig.12’ on slide 13 is based upon ‘化粧品基準/厚生省告示第331号(Standard for Cosmetics/MHLW Notification No.331).
- Q.What kind of documentation will be required when apply (cosmetic and quasi-drug and medicated drug)?
- A: ‘Ingredient’ and ‘claim’ are essentials for determination of category to apply.
- Q.Regarding the “Prohibited cases of product naming (cosmetic), Fig.10”, is it permitted to use the same name as that of “medicated cosmetics” (as it is a part of cosmetic according to the Fig.2)?
- A: Yes, it’s permitted to use the same name.
- Q.What is the difference between “cosmetic category 36 Prevent sunburn (Fig.16) ”, “37 Prevent skin splotches and freckles resulting from sunburn (Fig.16)” and “prevent sun spots and freckles (mediated cosmetic in Fig.19)” ? All of them seem very similar. Could you give me any example? Especially cosmetic 37 and medicated cosmetic in the effectiveness sun spots and freckles?
- A: As you point out, yes, their guideline is imperfect like this. However, in their notion, #37 is all about ‘UV protection’, not about that particular function as medicated cosmetics can promise.
- Q.I have a question regarding the import of products to Japan. If I look at figure 24 (business flow of exporting cosmetics to Japan), does it mean that the foreign manufacturer has to import products via a Japanese manufacturer?
- A: For importing cosmetics to Japan, it is necessary to have a Japan-based company which owns the license of primary distributor. This company (licensee) owes all responsible matters such as administrative procedures of local government. The licensee does not necessarily have to be the manufacturer, because the licensee does not do a “manufacturing activity”.
On the other hand, there is another rule that to introduce cosmetics to Japan, these cosmetics have to have a Japanese labeling. If you do the labeling in your own country and then export to Japan, that is OK. If any Japanese company handles the labeling process, that company has to have the license of manufacturing (category of “storage”, “display” and “packaging”). Also, if you temporarily store the products in Japanese warehouse before distributing to consumers, the same license is needed.
- Q.Fig 20: Approved medicated ingredients: After been approved, every company can use the ingredient in a Medical cosmetic. Or only the company that got the approval can use the ingredient?
- A: Only approved company can use the ingredient.
